
Selegiline
Monoamine oxidase inhibitor / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about selegiline?
Summarize this article for a 10 year old
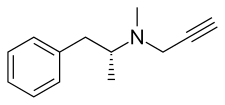
Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Zelapar, and Emsam among others, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder.[4][6][8][3] It has also been studied for a variety of other indications, but has not been formally approved for any other use.[20][21] The medication in the form licensed for depression has modest effectiveness for this condition that is similar to that of other antidepressants.[21][22][23] Selegiline is provided as a swallowed tablet or capsule[4][5] or an orally disintegrating tablet (ODT)[6][7] for Parkinson's disease and as a patch applied to skin for depression.[8][9]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /səˈlɛdʒɪliːn/ sə-LEJ-i-leen ("seh-LEH-ji-leen")[1][2] |
| Trade names | Eldepryl, Jumex, Zelapar, Emsam, Anipryl, others[3] |
| Other names | L-Deprenyl; L-Deprenil; L-Deprenalin; L-Deprenaline; L-E-250; L-Phenylisopropyl |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | • Oral (tablet, capsule)[4][5] • Buccal (ODTTooltip orally disintegrating tablet)[6][7] • Transdermal (patch)[8][9] |
| Drug class | Monoamine oxidase inhibitor; Catecholaminergic activity enhancer; Norepinephrine releasing agent; Antiparkinsonian; Antidepressant; Neuroprotective |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 4–10%[5][11][12] ODT: ~5–8× oral[13][7][14] Patch: 75%[9] |
| Protein binding | 85–90%[9][8][6] |
| Metabolism | Liver, other tissues (CYP2B6, CYP2C19, others)[5][15][9][16] |
| Metabolites | • Desmethylselegiline (DMS) • Levomethamphetamine (L-MA) • Levoamphetamine (L-A) |
| Elimination half-life | Oral: • S (single): 1.2–3.5 h[5] • S (multi): 7.7–9.7 h[5][12] • DMS (single): 2.2–3.8 h[5] • DMS (multi): 9.5 h[5] • L-MA: 14–21 h[5][7] • L-A: 16–18 h[5][7] ODT: • S (single): 1.3 h[6] • S (multi): 10 h[6] Patch: • S: 20 h[12][8] |
| Excretion | Urine (87%):[17][18][7][5][19] • L-MA: 20–63% • L-A: 9–26% • DMS: 1% • S: 0.01–0.03% Feces: 15%[17][7] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.109.269 |
| Chemical and physical data | |
| Formula | C13H17N |
| Molar mass | 187.286 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Levorotatory enantiomer |
| |
| |
| (verify) | |
Side effects of selegiline occurring more often than with placebo include insomnia, dry mouth, dizziness, nervousness, abnormal dreams, and application site reactions (with the patch form), among others.[21][22][24][4][8] At high doses, selegiline has the potential for dangerous food and drug interactions, such as the tyramine-related "cheese reaction" or hypertensive crisis and risk of serotonin syndrome.[9][25][5] However, doses within the approved clinical range appear to have little to no risk of these interactions.[9][25][5] In addition, the ODT and transdermal patch forms of selegiline have reduced risks of such interactions compared to the conventional oral form.[7][9] Selegiline has no known misuse potential or dependence liability and is not a controlled substance.[26][27][28][29][8]
Selegiline acts as a monoamine oxidase inhibitor (MAOI) and thereby increases levels of monoamine neurotransmitters in the brain.[30][11][25][5] At typical clinical doses used for Parkinson's disease, selegiline is a selective and irreversible inhibitor of monoamine oxidase B (MAO-B), increasing brain levels of dopamine.[30][11][25][5] At higher doses, it loses its specificity for MAO-B and also inhibits monoamine oxidase A (MAO-A), which increases serotonin and norepinephrine levels in the brain as well.[30][11][25][5] In addition to its MAOI activity, selegiline is a catecholaminergic activity enhancer (CAE) and enhances the impulse-mediated release of norepinephrine and dopamine in the brain.[31][32][33][34][25] This action may be mediated by TAAR1 agonism.[35][36][37] After administration, selegiline partially metabolizes into levomethamphetamine and levoamphetamine, which act as norepinephrine releasing agents (NRAs) and may contribute to its therapeutic and adverse effects.[38][28][39] The levels of these metabolites are much lower with the ODT and transdermal patch forms of selegiline.[7][9] Chemically, selegiline is a substituted amphetamine,[40] a derivative of methamphetamine,[40] and the purified levorotatory enantiomer of deprenyl (the racemic form).[41][20]
Deprenyl was discovered and studied in the early 1960s.[41][20] Subsequently, selegiline was purified from deprenyl and was studied and developed itself.[41] Selegiline was first introduced for medical use in Hungary in 1977.[42] It was subsequently approved in the United Kingdom in 1982 and in the United States in 1989.[42][43] The ODT was approved in the United States in 2006 and in the European Union in 2010, while the patch was introduced in the United States in 2006.[42][20] Selegiline was the first selective MAO-B inhibitor to be discovered and marketed.[13][44][45] In addition to its medical use, there has been interest in selegiline as a potential anti-aging drug and nootropic.[46] However, effects of this sort are controversial and uncertain.[47][48][49][50] Generic versions of selegiline are available in the case of the conventional oral form but not in the case of the ODT or transdermal patch forms.[51][52]