
Ganciclovir
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about ganciclovir?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Ganciclovir, sold under the brand name Cytovene among others, is an antiviral medication used to treat cytomegalovirus (CMV) infections.
Quick Facts Clinical data, Pronunciation ...
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɡænˈsaɪkləvɪər/ |
| Trade names | Cytovene; Cymevene; Vitrasert |
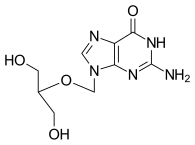
| Other names | gancyclovir; DHPG; 9-(1,3-dihydroxy-2-propoxymethyl)guanine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605011 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous, by mouth, intravitreal |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5% (oral) |
| Metabolism | guanylate kinase (CMV UL97 gene product) |
| Elimination half-life | 2.5–5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.155.403 |
| Chemical and physical data | |
| Formula | C9H13N5O4 |
| Molar mass | 255.234 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 250 °C (482 °F) (dec.) |
| |
| |
| (verify) | |
Close
Ganciclovir was patented in 1980 and approved for medical use in 1988.[4]