
Distillation
Method of separating mixtures / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about distillation?
Summarize this article for a 10 year old
Distillation, also classical distillation, is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances; the separation process is realized by way of the selective boiling of the mixture and the condensation of the vapors in a still.
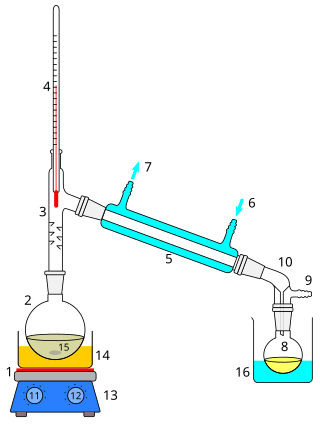
1: The heat source to boil the mixture
2: round-bottom flask containing the mixture to be boiled
3: the head of the still
4: mixture boling-point thermometre
5: the condenser of the still
6: the cooling-water inlet of the condenser
7: the cooling-water outlet of the condenser
8: the distillate-receiving flask
9: vacuum pump and gas inlet
10: the receiver of the still
11: the heat control for heating the mixture
12: stirring mechanism speed control
13: stirring mechanism and heating plate
14: heating bath (oil/sand) for the flask
15: the stirring mechanism (not shown, e.g. boiling chips or mechanical stirring machine)
16: the distillate-cooling water bath.[1]: 141–143
Dry distillation (thermolysis and pyrolysis) is the heating of solid materials to produce gases that condense either into fluid products or into solid products. The term dry distillation includes the separation processes of destructive distillation and of chemical cracking, breaking down large hydrocarbon molecules into smaller hydrocarbon molecules. Moreover, a partial distillation results in partial separations of the mixture's components, which process yields nearly-pure components; partial distillation also realizes partial separations of the mixture to increase the concentrations of selected components. In either method, the separation process of distillation exploits the differences in the relative volatility of the component substances of the heated mixture.
In the industrial applications of classical distillation, the term distillation is used as a unit of operation that identifies and denotes a process of physical separation, not a chemical reaction; thus an industrial installation that produces distilled beverages, is a distillery of alcohol. These are some applications of the chemical separation process that is distillation:
- Distilling fermented products to yield alcoholic beverages with a high content by volume of ethyl alcohol.
- Desalination to produce potable water and for medico-industrial applications.
- Crude oil stabilisation, a partial distillation to reduce the vapor pressure of crude oil, which thus is safe to store and to transport, and thereby reduces the volume of atmospheric emissions of volatile hydrocarbons.
- Fractional distillation used in the midstream operations of an oil refinery for producing fuels and chemical raw materials for livestock feed.[2][3][4]
- Cryogenic Air separation into the component gases — oxygen, nitrogen, and argon — for use as industrial gases.
- Chemical synthesis to separate impurities and unreacted materials.