Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
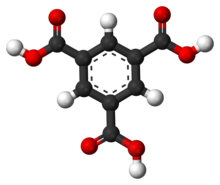
Trimesic acid, also known as benzene-1,3,5-tricarboxylic acid, is an organic compound with the formula C6H3(CO2H)3. It is one of three isomers of benzenetricarboxylic acid.[3] A colorless solid, trimesic acid has some commercial value as a precursor to some plasticizers.[4]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzene-1,3,5-tricarboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | TMA |
| 2053080 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.008.253 |
| EC Number |
|
| 51147 | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H6O6 | |
| Molar mass | 210.14034 |
| Acidity (pKa) | 3.12, 3.89, 4.70[1] |
| Hazards[2] | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Safety data sheet (SDS) | Oxford MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Trimesic acid can be combined with para-hydroxypyridine to make a water-based gel, stable up to 95 °C.[5]
Trimesic acid crystallizes from water to form a hydrogen-bonded hydrated network with wide unidimensional empty channels.[6][7]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.