Loading AI tools
Rock-forming minerals with predominantly silicate anions From Wikipedia, the free encyclopedia
Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most important class of minerals and make up approximately 90 percent of Earth's crust.[1][2][3]

In mineralogy, silica (silicon dioxide, SiO2) is usually considered a silicate mineral rather than an oxide mineral. Silica is found in nature as the mineral quartz, and its polymorphs.
On Earth, a wide variety of silicate minerals occur in an even wider range of combinations as a result of the processes that have been forming and re-working the crust for billions of years. These processes include partial melting, crystallization, fractionation, metamorphism, weathering, and diagenesis.

Living organisms also contribute to this geologic cycle. For example, a type of plankton known as diatoms construct their exoskeletons ("frustules") from silica extracted from seawater. The frustules of dead diatoms are a major constituent of deep ocean sediment, and of diatomaceous earth.[citation needed]
A silicate mineral is generally an inorganic compound consisting of subunits with the formula [SiO2+n]2n−. Although depicted as such, the description of silicates as anions is a simplification. Balancing the charges of the silicate anions are metal cations, Mx+. Typical cations are Mg2+, Fe2+, and Na+. The Si-O-M linkage between the silicates and the metals are strong, polar-covalent bonds. Silicate anions ([SiO2+n]2n−) are invariably colorless, or when crushed to a fine powder, white. The colors of silicate minerals arise from the metal component, commonly iron.
In most silicate minerals, silicon is tetrahedral, being surrounded by four oxides. The coordination number of the oxides is variable except when it bridges two silicon centers, in which case the oxide has a coordination number of two.
Some silicon centers may be replaced by atoms of other elements, still bound to the four corner oxygen corners. If the substituted atom is not normally tetravalent, it usually contributes extra charge to the anion, which then requires extra cations. For example, in the mineral orthoclase [KAlSi
3O
8]
n, the anion is a tridimensional network of tetrahedra in which all oxygen corners are shared. If all tetrahedra had silicon centers, the anion would be just neutral silica [SiO
2]
n. Replacement of one in every four silicon atoms by an aluminum atom results in the anion [AlSi
3O−
8]
n, whose charge is neutralized by the potassium cations K+
.
In mineralogy, silicate minerals are classified into seven major groups according to the structure of their silicate anion:[4][5]
| Major group | Structure | Chemical formula | Example |
|---|---|---|---|
| Nesosilicates | isolated silicon tetrahedra | [SiO4]4− | olivine, garnet, zircon... |
| Sorosilicates | double tetrahedra | [Si2O7]6− | epidote, melilite group |
| Cyclosilicates | rings | [SinO3n]2n− | beryl group, tourmaline group |
| Inosilicates | single chain | [SinO3n]2n− | pyroxene group |
| Inosilicates | double chain | [Si4nO11n]6n− | amphibole group |
| Phyllosilicates | sheets | [Si2nO5n]2n− | micas and clays |
| Tectosilicates | 3D framework | [AlxSiyO(2x+2y)]x− | quartz, feldspars, zeolites |
Tectosilicates can only have additional cations if some of the silicon is replaced by an atom of lower valence such as aluminum. Al for Si substitution is common.


Nesosilicates (from Greek νῆσος nēsos 'island'), or orthosilicates, have the orthosilicate ion, present as isolated (insular) [SiO4]4− tetrahedra connected only by interstitial cations. The Nickel–Strunz classification is 09.A –examples include:



Sorosilicates (from Greek σωρός sōros 'heap, mound') have isolated pyrosilicate anions Si
2O6−
7, consisting of double tetrahedra with a shared oxygen vertex—a silicon:oxygen ratio of 2:7. The Nickel–Strunz classification is 09.B. Examples include:



Cyclosilicates (from Greek κύκλος kýklos 'circle'), or ring silicates, have three or more tetrahedra linked in a ring. The general formula is (SixO3x)2x−, where one or more silicon atoms can be replaced by other 4-coordinated atom(s). The silicon:oxygen ratio is 1:3. Double rings have the formula (Si2xO5x)2x− or a 2:5 ratio. The Nickel–Strunz classification is 09.C. Possible ring sizes include:
Some example minerals are:
The ring in axinite contains two B and four Si tetrahedra and is highly distorted compared to the other 6-member ring cyclosilicates.
Inosilicates (from Greek ἴς is [genitive: ἰνός inos] 'fibre'), or chain silicates, have interlocking chains of silicate tetrahedra with either SiO3, 1:3 ratio, for single chains or Si4O11, 4:11 ratio, for double chains. The Nickel–Strunz classification is 09.D – examples include:
Phyllosilicates (from Greek φύλλον phýllon 'leaf'), or sheet silicates, form parallel sheets of silicate tetrahedra with Si2O5 or a 2:5 ratio. The Nickel–Strunz classification is 09.E. All phyllosilicate minerals are hydrated, with either water or hydroxyl groups attached.

Examples include:
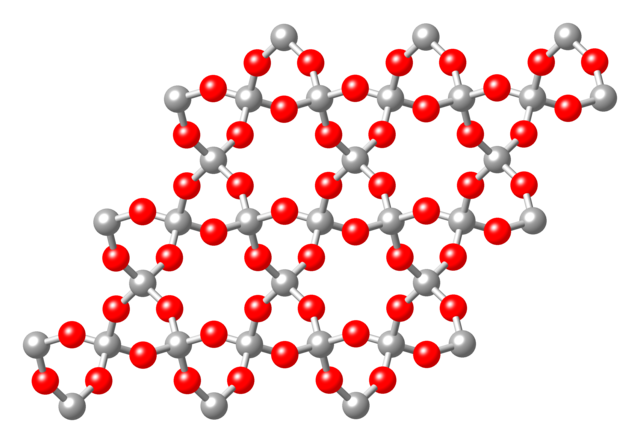



Tectosilicates, or "framework silicates," have a three-dimensional framework of silicate tetrahedra with SiO2 in a 1:2 ratio. This group comprises nearly 75% of the crust of the Earth.[6] Tectosilicates, with the exception of the quartz group, are aluminosilicates. The Nickel–Strunz classifications are 09.F and 09.G, 04.DA (Quartz/ silica family). Examples include:
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.