Loading AI tools
Hearing loss caused by an inner ear or vestibulocochlear nerve defect From Wikipedia, the free encyclopedia
Sensorineural hearing loss (SNHL) is a type of hearing loss in which the root cause lies in the inner ear, sensory organ (cochlea and associated structures), or the vestibulocochlear nerve (cranial nerve VIII). SNHL accounts for about 90% of reported hearing loss.[citation needed] SNHL is usually permanent and can be mild, moderate, severe, profound, or total. Various other descriptors can be used depending on the shape of the audiogram, such as high frequency, low frequency, U-shaped, notched, peaked, or flat.
| Sensorineural hearing loss | |
|---|---|
 | |
| Cross section of the cochlea. | |
| Specialty | Otorhinolaryngology |
Sensory hearing loss often occurs as a consequence of damaged or deficient cochlear hair cells.[disputed – discuss] Hair cells may be abnormal at birth or damaged during the lifetime of an individual. There are both external causes of damage, including infection, and ototoxic drugs, as well as intrinsic causes, including genetic mutations. A common cause or exacerbating factor in SNHL is prolonged exposure to environmental noise, or noise-induced hearing loss. Exposure to a single very loud noise such as a gun shot or bomb blast can cause noise-induced hearing loss. Using headphones at high volume over time, or being in loud environments regularly, such as a loud workplace, sporting events, concerts, and using noisy machines can also be a risk for noise-induced hearing loss.
Neural, or "retrocochlear", hearing loss occurs because of damage to the cochlear nerve (CVIII). This damage may affect the initiation of the nerve impulse in the cochlear nerve or the transmission of the nerve impulse along the nerve into the brainstem.
Most cases of SNHL present with a gradual deterioration of hearing thresholds occurring over years to decades. In some, the loss may eventually affect large portions of the frequency range. It may be accompanied by other symptoms such as ringing in the ears (tinnitus) and dizziness or lightheadedness (vertigo). The most common kind of sensorineural hearing loss is age-related (presbycusis), followed by noise-induced hearing loss (NIHL).
Frequent symptoms of SNHL are loss of acuity in distinguishing foreground voices against noisy backgrounds, difficulty understanding on the telephone, some kinds of sounds seeming excessively loud or shrill, difficulty understanding some parts of speech (fricatives and sibilants), loss of directionality of sound (especially with high frequency sounds), perception that people mumble when speaking, and difficulty understanding speech. Similar symptoms are also associated with other kinds of hearing loss; audiometry or other diagnostic tests are necessary to distinguish sensorineural hearing loss.
Identification of sensorineural hearing loss is usually made by performing a pure tone audiometry (an audiogram) in which bone conduction thresholds are measured. Tympanometry and speech audiometry may be helpful. Testing is performed by an audiologist.
There is no proven or recommended treatment or cure for SNHL; management of hearing loss is usually by hearing strategies and hearing aids. In cases of profound or total deafness, a cochlear implant is a specialised device that may restore a functional level of hearing. SNHL is at least partially preventable by avoiding environmental noise, ototoxic chemicals and drugs, and head trauma, and treating or inoculating against certain triggering diseases and conditions like meningitis.
Since the inner ear is not directly accessible to instruments, identification is by patient report of the symptoms and audiometric testing. Of those who present to their doctor with sensorineural hearing loss, 90% report having diminished hearing, 57% report having a plugged feeling in ear, and 49% report having ringing in ear (tinnitus). About half report vestibular (vertigo) problems.[citation needed]
For a detailed exposition of symptoms useful for screening, a self-assessment questionnaire was developed by the American Academy of Otolaryngology, called the Hearing Handicap Inventory for Adults (HHIA). It is a 25-question survey of subjective symptoms.[1]
Sensorineural hearing loss may be genetic or acquired (i.e. as a consequence of disease, noise, trauma, etc.). People may have a hearing loss from birth (congenital) or the hearing loss may come on later. Many cases are related to old age (age-related).[citation needed]
Hearing loss can be inherited. More than 40 genes have been implicated in the cause of deafness.[2] There are 300 syndromes with related hearing loss, and each syndrome may have causative genes.[citation needed]
Recessive, dominant, X-linked, or mitochondrial genetic mutations can affect the structure or metabolism of the inner ear. Some may be single point mutations, whereas others are due to chromosomal abnormalities. Some genetic causes give rise to a late onset hearing loss. Mitochondrial mutations can cause SNHL i.e. m.1555A>G, which makes the individual sensitive to the ototoxic effects of aminoglycoside antibiotics.
Progressive age-related loss of hearing acuity or sensitivity can start as early as age 18, primarily affecting the high frequencies, and men more than women.[6] Such losses may not become apparent until much later in life. Presbycusis is by far the dominant cause of sensorineural hearing loss in industrialized societies. A study conducted in Sudan, with a population free from loud noise exposures, found significantly less cases of hearing loss when compared with age-matched cases from an industrialized country.[7] Similar findings were reported by a study conducted of a population from Easter island, which reported worse hearing among those that spent time in industrialized countries when compared with those that never left the island.[8] Researchers have argued that factors other than differences in noise exposure, such as genetic make up, might also have contributed to the findings.[9] Hearing loss that worsens with age but is caused by factors other than normal aging, such as noise-induced hearing loss, is not presbycusis, although differentiating the individual effects of multiple causes of hearing loss can be difficult. One in three persons have significant hearing loss by age 65; by age 75, one in two. Age-related hearing loss is neither preventable nor reversible.
Most people living in modern society have some degree of progressive sensorineural (i.e. permanent) noise-induced hearing loss (NIHL) resulting from overloading and damaging the sensory or neural apparatus of hearing in the inner ear.[citation needed] NIHL is typically a drop-out or notch centered at 4000 Hz. Both intensity (SPL) and duration of exposure, and repetitive exposure to unsafe levels of noise contribute to cochlear damage that results in hearing loss. The louder the noise is, the shorter the safe amount of exposure is. NIHL can be either permanent or temporary, called a threshold shift. Unsafe levels of noise can be as little as 70 dB (about twice as loud as normal conversation) if there is prolonged (24-hour) or continuous exposure. 125 dB (a loud rock concert is ~120 dB) is the pain level; sounds above this level cause instant and permanent ear damage.[citation needed]
Noise and ageing are the primary causes of presbycusis, or age-related hearing loss, the most common kind of hearing loss in industrial society.[10] [citation needed] The dangers of environmental and occupational noise exposure are widely recognized. Numerous national and international organizations have established standards for safe levels of exposure to noise in industry, the environment, military, transportation, agriculture, mining and other areas.[Note 1] Sound intensity or sound pressure level (SPL) is measured in decibels (dB). For reference:
| db Level | Example |
|---|---|
| 45 dB | Ambient noise level around the home |
| 60 dB | Quiet office |
| 60–65 dB | Normal conversation |
| 70 dB | City street noise at 25'[clarification needed] or average TV audio |
| 80 dB | Noisy office |
| 95–104 dB | Nightclub dance floor |
| 120 dB | Close by thunder or a loud rock concert |
| 150–160 dB | Gunshot from a handheld gun |
An increase of 6 dB represents a doubling of the SPL, or energy of the sound wave, and therefore its propensity to cause ear damage. Because human ears hear logarithmically, not linearly, it takes an increase of 10 dB to produce a sound that is perceived to be twice as loud. Ear damage due to noise is proportional to sound intensity, not perceived loudness, so it's misleading to rely on subjective perception of loudness as an indication of the risk to hearing, i.e. it can significantly underestimate the danger.
While the standards differ moderately in levels of intensity and duration of exposure considered safe, some guidelines can be derived.[Note 2]
The safe amount of exposure is reduced by a factor of 2 for every exchange rate (3 dB for NIOSH standard or 5 dB for OSHA standard) increase in SPL. For example, the safe daily exposure amount at 85 dB (90 dB for OSHA) is 8 hours, while the safe exposure at 94 dB(A) (nightclub level) is only 1 hour. Noise trauma can also cause a reversible hearing loss, called a temporary threshold shift. This typically occurs in individuals who are exposed to gunfire or firecrackers, and hear ringing in their ears after the event (tinnitus).
Some over-the-counter as well as prescription drugs and certain industrial chemicals are ototoxic. Exposure to these can result in temporary or permanent hearing loss.
Some medications cause irreversible damage to the ear, and are limited in their use for this reason. The most important group is the aminoglycosides (main member gentamicin). A rare mitochondrial mutation, m.1555A>G, can increase an individual's susceptibility to the ototoxic effect of aminoglycosides. Long term hydrocodone (Vicodin) abuse is known to cause rapidly progressing sensorineural hearing loss, usually without vestibular symptoms. Methotrexate, a chemotherapy agent, is also known to cause hearing loss. In most cases hearing loss does not recover when the drug is stopped. Paradoxically, methotrexate is also used in the treatment of autoimmune-induced inflammatory hearing loss.[citation needed]
Various other medications may reversibly degrade hearing. This includes loop diuretics, sildenafil (Viagra), high or sustained dosing of NSAIDs (aspirin, ibuprofen, naproxen, and various prescription drugs: celecoxib, etc.), quinine, and macrolide antibiotics (erythromycin, etc.). Cytotoxic agents such as carboplatinum, used to treat malignancies can give rise to a dose dependent SNHL, as can drugs such as desferrioxamine, used for haematological disorders such as thalassaemia; patients prescribed these drugs need to have hearing monitored.[citation needed]
Prolonged or repeated environmental or work-related exposure to ototoxic chemicals can also result in sensorineural hearing loss. Some of these chemicals are:
There can be damage either to the ear itself or to the central auditory pathways that process the information conveyed by the ears. People who sustain head injury are susceptible to hearing loss or tinnitus, either temporary or permanent. Contact sports like football (U.S. NFL), hockey and cricket have a notable incidence of head injuries (concussions). In one survey of retired NFL players, all of whom reported one or more concussions during their playing careers, 25% had hearing loss and 50% had tinnitus.[citation needed]
These are much more common in premature babies, particularly those under 1500 g at birth. Premature birth can be associated with problems that result in sensorineural hearing loss such as anoxia or hypoxia (poor oxygen levels), jaundice, intracranial haemorrhages, meningitis. Fetal alcohol syndrome is reported to cause hearing loss in up to 64% of infants born to alcoholic mothers, from the ototoxic effect on the developing fetus, plus malnutrition during pregnancy from the excess alcohol intake.
Iodine deficiency and endemic hypothyroidism are associated with hearing loss.[12] If a pregnant mother has insufficient iodine intake during pregnancy it affects the development of the inner ear in the foetus leading to sensorineural deafness. This occurs in certain areas of the world, such as the Himalayas, where iodine is deficient in the soil and thus the diet. In these areas there is a high incidence of endemic goitre. This cause of deafness is prevented by adding iodine to salt.
Brain stroke in a region affecting auditory function such as a posterior circulation infarct has been associated with deafness.
Sensory hearing loss is caused by abnormal structure or function of the hair cells of the organ of Corti in the cochlea.[disputed – discuss] Neural hearing impairments are consequent upon damage to the eighth cranial nerve (the vestibulocochlear nerve) or the auditory tracts of the brainstem. If higher levels of the auditory tract are affected this is known as central deafness. Central deafness may present as sensorineural deafness but should be distinguishable from the history and audiological testing.
Hearing impairment may be associated with damage to the hair cells in the cochlea. Sometimes there may be complete loss of function of inner hair cells (IHCs) over a certain region of the cochlea; this is called a "dead region". The region can be defined in terms of the range of characteristic frequencies (CFs) of the IHCs and/or neurons immediately adjacent to the dead region.

Outer hair cells (OHCs) contribute to the structure of the Organ of Corti, which is situated between the basilar membrane and the tectorial membrane within the cochlea (See Figure 3). The tunnel of corti, which runs through the Organ of Corti, divides the OHCs and the inner hair cells (IHCs). OHCs are connected to the reticular laminar and the Deiters’ cells. There are roughly twelve thousand OHCs in each human ear, and these are arranged in up to five rows. Each OHC has small tufts of 'hairs', or cilia, on their upper surface known as stereocilia, and these are also arranged into rows which are graded in height. There are approximately 140 stereocilia on each OHC.[13]
The fundamental role of the OHCs and the IHCs is to function as sensory receptors. The main function of the IHCs is to transmit sound information via afferent neurons. They do this by transducing mechanical movements or signals into neural activity. When stimulated, the stereocilia on the IHCs move, causing a flow of electric current to pass through the hair cells. This electric current creates action potentials within the connected afferent neurons.
OHCs are different in that they actually contribute to the active mechanism of the cochlea. They do this by receiving mechanical signals or vibrations along the basilar membrane, and transducing them into electrochemical signals. The stereocilia found on OHCs are in contact with the tectorial membrane. Therefore, when the basilar membrane moves due to vibrations, the stereocilia bend. The direction in which they bend, dictates the firing rate of the auditory neurons connected to the OHCs.[14]
The bending of the stereocilia towards the basal body of the OHC causes excitation of the hair cell. Thus, an increase in firing rate of the auditory neurons connected to the hair cell occurs. On the other hand, the bending of the stereocilia away from the basal body of the OHC causes inhibition of the hair cell. Thus, a decrease in firing rate of the auditory neurons connected to the hair cell occurs. OHCs are unique in that they are able to contract and expand (electromotility). Therefore, in response to the electrical stimulations provided by the efferent nerve supply, they can alter in length, shape and stiffness. These changes influence the response of the basilar membrane to sound.[13][14] It is therefore clear that the OHCs play a major role in the active processes of the cochlea.[13] The main function of the active mechanism is to finely tune the basilar membrane, and provide it with a high sensitivity to quiet sounds. The active mechanism is dependent on the cochlea being in good physiological condition. However, the cochlea is very susceptible to damage.[14]
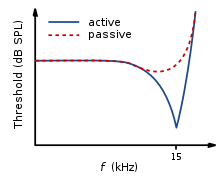
SNHL is most commonly caused by damage to the OHCs and the IHCs.[disputed – discuss] There are two methods by which they might become damaged. Firstly, the entire hair cell might die. Secondly, the stereocilia might become distorted or destroyed. Damage to the cochlea can occur in several ways, for example by viral infection, exposure to ototoxic chemicals, and intense noise exposure. Damage to the OHCs results in either a less effective active mechanism, or it may not function at all. OHCs contribute to providing a high sensitivity to quiet sounds at a specific range of frequencies (approximately 2–4 kHz). Thus, damage to the OHCs results in the reduction of sensitivity of the basilar membrane to weak sounds. Amplification to these sounds is therefore required, in order for the basilar membrane to respond efficiently. IHCs are less susceptible to damage in comparison to the OHCs. However, if they become damaged, this will result in an overall loss of sensitivity.[14]
The traveling wave along the basilar membrane peaks at different places along it, depending on whether the sound is low or high frequency. Due to the mass and stiffness of the basilar membrane, low frequency waves peak in the apex, while high frequency sounds peak in the basal end of the cochlea.[13] Therefore, each position along the basilar membrane is finely tuned to a particular frequency. These specifically tuned frequencies are referred to as characteristic frequencies (CF).[14]
If a sound entering the ear is displaced from the characteristic frequency, then the strength of response from the basilar membrane will progressively lessen. The fine tuning of the basilar membrane is created by the input of two separate mechanisms. The first mechanism being a linear passive mechanism, which is dependent on the mechanical structure of the basilar membrane and its surrounding structures. The second mechanism is a non-linear active mechanism, which is primarily dependent on the functioning of the OHCs, and also the general physiological condition of the cochlea itself. The base and apex of the basilar membrane differ in stiffness and width, which cause the basilar membrane to respond to varying frequencies differently along its length. The base of the basilar membrane is narrow and stiff, resulting in it responding best to high frequency sounds. The apex of the basilar membrane is wider and much less stiff in comparison to the base, causing it to respond best to low frequencies.[14]
This selectivity to certain frequencies can be illustrated by neural tuning curves. These demonstrate the frequencies a fiber responds to, by showing threshold levels (dB SPL) of auditory nerve fibers as a function of different frequencies. This demonstrates that auditory nerve fibers respond best, and hence have better thresholds at the fiber's characteristic frequency and frequencies immediately surrounding it. The basilar membrane is said to be ‘sharply tuned’ due to the sharp V-shaped curve, with its ‘tip’ centered at the auditory fibers characteristic frequency. This shape shows how few frequencies a fiber responds to. If it were a broader ‘V’ shape, it would be responding to more frequencies (See Figure 4).[13]


A normal neural tuning curve is characterised by a broadly tuned low frequency ‘tail’, with a finely tuned middle frequency ‘tip’. However, where there is partial or complete damage to the OHCs, but with unharmed IHCs, the resulting tuning curve would show the elimination of sensitivity at the quiet sounds. I.e. where the neural tuning curve would normally be most sensitive (at the ‘tip’) (See Figure 5).[14]
Where both the OHCs and the IHCs are damaged, the resulting neural tuning curve would show the elimination of sensitivity at the ‘tip'. However, due to IHC damage, the whole tuning curve becomes raised, giving a loss of sensitivity across all frequencies (See Figure 6). It is only necessary for the first row of OHCs to be damaged for the elimination of the finely tuned ‘tip’ to occur. This supports the idea that the incidence of OHC damage and thus a loss of sensitivity to quiet sounds, occurs more than IHC loss.[14]
When the IHCs or part of the basilar membrane are damaged or destroyed, so that they no longer function as transducers, the result is a ‘dead region’. Dead regions can be defined in terms of the characteristic frequencies of the IHC, related to the specific place along the basilar membrane where the dead region occurs. Assuming that there has been no shift in the characteristic frequencies relating to certain regions of the basilar membrane, due to the damage of OHCs. This often occurs with IHC damage. Dead regions can also be defined by the anatomical place of the non-functioning IHC (such as an “apical dead region”), or by the characteristic frequencies of the IHC adjacent to the dead region.[15]
Dead regions affect audiometric results, but perhaps not in the way expected. For example, it may be expected that thresholds would not be obtained at the frequencies within the dead region, but would be obtained at frequencies adjacent to the dead region. Therefore, assuming normal hearing exists around the dead region, it would produce an audiogram that has a dramatically steep slope between the frequency where a threshold is obtained, and the frequency where a threshold cannot be obtained due to the dead region.[15]


However, it appears that this is not the case. Dead regions cannot be clearly found via PTA audiograms. This may be because although the neurons innervating the dead region, cannot react to vibration at their characteristic frequency. If the basilar membrane vibration is large enough, neurons tuned to different characteristic frequencies such as those adjacent to the dead region, will be stimulated due to the spread of excitation. Therefore, a response from the patient at the test frequency will be obtained. This is referred to as “off-place listening”, and is also known as ‘off-frequency listening’. This will lead to a false threshold being found. Thus, it appears a person has better hearing than they actually do, resulting in a dead region being missed. Therefore, using PTA alone, it is impossible to identify the extent of a dead region (See Figure 7 and 8).[15]
Consequently, how much is an audiometric threshold affected by a tone with its frequency within a dead region? This depends on the location of the dead region. Thresholds at low frequency dead regions, are more inaccurate than those at higher frequency dead regions. This has been attributed to the fact that excitation due to vibration of the basilar membrane spreads upwards from the apical regions of the basilar membrane, more than excitation spreads downwards from higher frequency basal regions of the cochlea. This pattern of the spread of excitation is similar to the ‘upward spread of masking’ phenomenon. If the tone is sufficiently loud to produce enough excitation at the normally functioning area of the cochlea, so that it is above that areas threshold. The tone will be detected, due to off-frequency listening which results in a misleading threshold.[15]
To help to overcome the issue of PTA producing inaccurate thresholds within dead regions, masking of the area beyond the dead region that is being stimulated can be used. This means that the threshold of the responding area is sufficiently raised, so that it cannot detect the spread of excitation from the tone. This technique has led to the suggestion that a low frequency dead region may be related to a loss of 40-50 dB.[16][17] However, as one of the aims of PTA is to determine whether or not there is a dead region, it may be difficult to assess which frequencies to mask without the use of other tests.[15]
Based on research it has been suggested that a low frequency dead region may produce a relatively flat loss, or a very gradually sloping loss towards the higher frequencies. As the dead region will be less detectable due to the upward spread of excitation. Whereas, there may be a more obvious steeply sloping loss at high frequencies for a high frequency dead region. Although it is likely that the slope represents the less pronounced downward spread of excitation, rather than accurate thresholds for those frequencies with non-functioning hair cells. Mid-frequency dead regions, with a small range, appear to have less effect on the patient's ability to hear in everyday life, and may produce a notch in the PTA thresholds.[15] Although it is clear that PTA is not the best test to identify a dead region.[18]
This section possibly contains unsourced predictions, speculative material, or accounts of events that might not occur. Information must be verifiable and based on reliable published sources. (November 2015) |

Although some debate continues regarding the reliability of such tests,[19] it has been suggested [weasel words]that psychoacoustic tuning curves (PTCs) and threshold-equalising noise (TEN) results may be useful in detecting dead regions, rather than PTA. PTCs are similar to neural tuning curves. They illustrate the level of a masker (dB SPL) tone at threshold, as a function of deviation from center frequency (Hz).[13] They are measured by presenting a fixed low intensity pure tone while also presenting a narrow-band masker, with a varying center frequency. The masker level is varied, so that the level of masker needed to just mask the test signal is found for the masker at each center frequency. The tip of the PTC is where the masker level needed to just mask the test signal is the lowest. For normal hearing people this is when the masker center frequency is closest to the frequency of the test signal (See Figure 9).[18]
In the case of dead regions, when the test signal lies within the boundaries of a dead region, the tip of the PTC will be shifted to the edge of the dead region, to the area that is still functioning and detecting the spread of excitation from the signal. In the case of a low frequency dead region, the tip is shifted upwards indicating a low frequency dead region starting at the tip of the curve. For a high frequency dead region, the tip is shifted downwards from the signal frequency to the functioning area below the dead region.[18] However, the traditional method of obtaining PTCs is not practical for clinical use, and it has been argued[weasel words] that TENs are not accurate enough.[18][19] A fast method for finding PTCs has been developed and it may provide the solution. However, more research to validate this method is required, before it can be accepted clinically.
Audiogram configurations are not good indicators of how a dead region will affect a person functionally, mainly due to individual differences.[14] For example, a sloping audiogram is often present with a dead region, due to the spread of excitation. However, the individual may well be affected differently from someone with a corresponding sloped audiogram caused by partial damage to hair cells rather than a dead region. They will perceive sounds differently, yet the audiogram suggests that they have the same degree of loss. Huss and Moore investigated how hearing impaired patients perceive pure tones, and found that they perceive tones as noisy and distorted, more (on average) than a person without a hearing impairment. However, they also found that the perception of tones as being like noise, was not directly related to frequencies within the dead regions, and was therefore not an indicator of a dead region. This therefore suggests that audiograms, and their poor representation of dead regions, are inaccurate predictors of a patient's perception of pure tone quality.[20]
Research by Kluk and Moore has shown that dead regions may also affect the patient's perception of frequencies beyond the dead regions. There is an enhancement in the ability to distinguish between tones that differ very slightly in frequency, in regions just beyond the dead regions compared to tones further away. An explanation for this may be that cortical re-mapping has occurred. Whereby, neurons which would normally be stimulated by the dead region, have been reassigned to respond to functioning areas near it. This leads to an over-representation of these areas, resulting in an increased perceptual sensitivity to small frequency differences in tones.[21]
Before examination, a case history provides guidance about the context of the hearing loss.
Direct examination of the external canal and tympanic membrane (ear drum) with an otoscope, a medical device inserted into the ear canal that uses light to examine the condition of the external ear and tympanic membrane, and middle ear through the semi-translucent membrane.
Differential testing is most useful when there is unilateral hearing loss, and distinguishes conductive from sensorineural loss. These are conducted with a low frequency tuning fork, usually 512 Hz, and contrast measures of air and bone conducted sound transmission.
Table 1. A table comparing sensorineural to conductive hearing loss
| Criteria | Sensorineural hearing loss | Conductive hearing loss |
| Anatomical site | Inner ear, cranial nerve VIII, or central processing centers | Middle ear (ossicular chain), tympanic membrane, or external ear |
| Weber test | Sound localizes to normal ear in unilateral SNHL | Sound localizes to affected ear (ear with conductive loss) in unilateral cases |
| Rinne test | Positive Rinne; air conduction > bone conduction (both air and bone conduction are decreased equally, but the difference between them is unchanged). | Negative Rinne; bone conduction > air conduction (bone/air gap) |
Other, more complex, tests of auditory function are required to distinguish the different types of hearing loss. Bone conduction thresholds can differentiate sensorineural hearing loss from conductive hearing loss. Other tests, such as oto-acoustic emissions, acoustic stapedial reflexes, speech audiometry and evoked response audiometry are needed to distinguish sensory, neural and auditory processing hearing impairments.
A tympanogram is the result of a test with a tympanometer. It tests the function of the middle ear and mobility of the eardrum. It can help identify conductive hearing loss due to disease of the middle ear or eardrum from other kinds of hearing loss including SNHL.
An audiogram is the result of a hearing test. The most common type of hearing test is pure tone audiometry (PTA). It charts the thresholds of hearing sensitivity at a selection of standard frequencies between 250 and 8000 Hz. There is also high frequency pure tone audiometry which tests frequencies from 8000 to 20,000 Hz. PTA can be used to differentiate between conductive hearing loss, sensorineural hearing loss and mixed hearing loss. A hearing loss can be described by its degree i.e. mild, moderate, severe or profound, or by its shape i.e. high frequency or sloping, low frequency or rising, notched, U-shaped or 'cookie-bite', peaked or flat.
There are also other kinds of audiometry designed to test hearing acuity rather than sensitivity (speech audiometry), or to test auditory neural pathway transmission (evoked response audiometry).
MRI scans can be used to identify gross structural causes of hearing loss. They are used for congenital hearing loss when changes to the shape of the inner ear or nerve of hearing may help diagnosis of the cause of the hearing loss. They are also useful in cases where a tumour is suspected or to determine the degree of damage in a hearing loss caused by bacterial infection or auto-immune disease. Scanning is of no value in age-related deafness.
Presbycusis is the leading cause of SNHL and is progressive and nonpreventable, and at this time, we do not have either somatic or gene therapy to counter heredity-related SNHL. But other causes of acquired SNHL are largely preventable, especially nosocusis type causes. This would involve avoiding environmental noise, and traumatic noise such as rock concerts and nightclubs with loud music. Use of noise attenuation measures like ear plugs is an alternative, as well as learning about the noise levels one is exposed to. Currently, several accurate sound level measurement apps exist. Reducing exposure time can also help manage risk from loud exposures.

Treatment modalities fall into three categories: pharmacological, surgical, and management. As SNHL is a physiologic degradation and considered permanent, there are as of this time, no approved or recommended treatments.
There have been significant advances in identification of human deafness genes and elucidation of their cellular mechanisms as well as their physiological function in mice.[22][23] Nevertheless, pharmacological treatment options are very limited and clinically unproven.[24] Such pharmaceutical treatments as are employed are palliative rather than curative, and addressed to the underlying cause if one can be identified, in order to avert progressive damage.
Profound or total hearing loss may be amenable to management by cochlear implants, which stimulate cochlear nerve endings directly. A cochlear implant is surgical implantation of a battery powered electronic medical device in the inner ear. Unlike hearing aids, which make sounds louder, cochlear implants do the work of damaged parts of the inner ear (cochlea) to provide sound signals to the brain. These consist of both internal implanted electrodes and magnets and external components.[25] The quality of sound is different than natural hearing but may enable the recipient to better recognize speech and environmental sounds. Because of risk and expense, such surgery is reserved for cases of severe and disabling hearing impairment
Management of sensorineural hearing loss involves employing strategies to support existing hearing such as lip-reading, enhanced communication etc. and amplification using hearing aids. Hearing aids are specifically tuned to the individual hearing loss to give maximum benefit.
Hair cell regeneration using stem cell and gene therapy is years or decades away from being clinically feasible.[28] However, studies are currently underway on the subject, with the first FDA-approved trial beginning in February 2012.[29]
Sudden sensorineural hearing loss (SSHL or SSNHL), commonly known as sudden deafness, occurs as an unexplained, rapid loss of hearing—usually in one ear—either at once or over several days. Nine out of ten people with SSHL lose hearing in only one ear. It should be considered a medical emergency. Delaying diagnosis and treatment may render treatment less effective or ineffective.
Experts estimate that SSHL strikes one person per 100 every year, typically adults in their 40s and 50s. The actual number of new cases of SSHL each year could be much higher because the condition often goes undiagnosed.
Many people notice that they have SSHL when they wake up in the morning. Others first notice it when they try to use the deafened ear, such as when they use a phone. Still others notice a loud, alarming "pop" just before their hearing disappears. People with sudden deafness often become dizzy, have ringing in their ears (tinnitus), or both.
SSHL is diagnosed via pure tone audiometry. If the test shows a loss of at least 30 dB in three adjacent frequencies, the hearing loss is diagnosed as SSHL. For example, a hearing loss of 30 dB would make conversational speech sound more like a whisper.
Only 10 to 15 percent of the cases diagnosed as SSHL have an identifiable cause. Most cases are classified as idiopathic, also called sudden idiopathic hearing loss (SIHL) and idiopathic sudden sensorineural hearing loss (ISSHL or ISSNHL)[30][31] The majority of evidence points to some type of inflammation in the inner ear as the most common cause of SSNHL.
Hearing loss completely recovers in around 35–39% of patients with SSNHL, usually within one to two weeks from onset.[34] Steroid treatment within seven days, a lower initial severity of hearing loss, the absence of vertigo, younger patient age, and a history of cardiovascular disease are all associated with complete hearing recovery.[35] Eighty-five percent of those who receive treatment from an otolaryngologist (sometimes called an ENT surgeon) will recover some of their hearing.[citation needed]
General hearing loss affects close to 10% of the global population.[42] In the United States alone, it is expected that 13.5 million Americans have sensorineural hearing loss. Of those with sensorineural hearing loss, approximately 50% are congenitally related. The other 50% are due to maternal or fetal infections, post-natal infections, viral infections due to rubella or cytomegalovirus, ototoxic drugs,[43] exposure to loud sounds, severe head trauma, and premature births [44]
Of the genetically related sensorineural hearing loss cases, 75% are autosomal recessive, 15-20% autosomal dominant, and 1-3% sex-linked. While the specific gene and protein is still unknown, mutations in the connexin 26 gene near the DFNB1 locus of chromosome 13[45] are thought to account for most of the autosomal recessive genetic-related sensorineural hearing loss [44]
At least 8.5 per 1000 children younger than age 18 have sensorineural hearing loss. General hearing loss is proportionally related to age. At least 314 per 1000 people older than age 65 have hearing loss. Several risk factors for sensorineural hearing loss have been studied over the past decade. Osteoporosis, stapedectomy surgery, pneumococcal vaccinations, mobile phone users, and hyperbilirubinemia at birth are among some of the known risk factors.
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.