Top Qs
Timeline
Chat
Perspective
Myo-Inositol trispyrophosphate
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
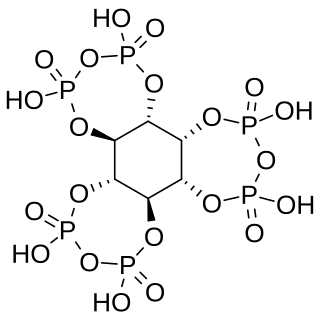
myo-Inositol trispyrophosphate (ITPP) is an inositol phosphate, a pyrophosphate, a drug candidate, and a putative performance-enhancing substance, which exerts its biological effects by increasing tissue oxygenation.[1]
Remove ads
Remove ads
Chemistry
ITPP is a pyrophosphate derivative of phytic acid with the molecular formula C6H12O21P6.[1]
Biological effects
Summarize
Perspective
ITPP is a membrane-permeant allosteric regulator of hemoglobin that mildly reduces its oxygen-binding affinity, which shifts the oxygen-hemoglobin dissociation curve to the right and thereby increases oxygen release from the blood into tissue.[1] Phytic acid, in contrast, is not membrane-permeant due to its charge distribution.[1]
Rodent studies in vivo demonstrated increased tissue oxygenation and dose-dependent increases in endurance during physical exercise, in both healthy mice and transgenic mice expressing a heart failure phenotype.[1]
The substance is believed to have a high potential for use in athletic doping, and liquid chromatography–mass spectrometry tests have been developed to detect ITPP in urine tests.[2] Its use as a performance-enhancing substance in horse racing has also been suspected and similar tests have been developed for horses[3]
ITPP has been studied for potential adjuvant use in the treatment of cancer in conjunction with chemotherapy, due to its effects in reducing tissue hypoxia.[4] Human clinical trials were registered in 2014 under the compound number OXY111A.[5] The substance has also been examined in the context of other illnesses involving hypoxia, such as cardiovascular disease and dementia[2]
Remove ads
See also
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads