Sorption is a physical and chemical process by which one substance becomes attached to another. Specific cases of sorption are treated in the following articles:
- Absorption
- "the incorporation of a substance in one state into another of a different state"[1] (e.g., liquids being absorbed by a solid or gases being absorbed by a liquid);
- Adsorption
- The physical adherence or bonding of ions and molecules onto the surface of another phase (e.g., reagents adsorbed to a solid catalyst surface);
- Ion exchange
- An exchange of ions between two electrolytes or between an electrolyte solution and a complex.

The reverse of sorption is desorption.
Sorption rate
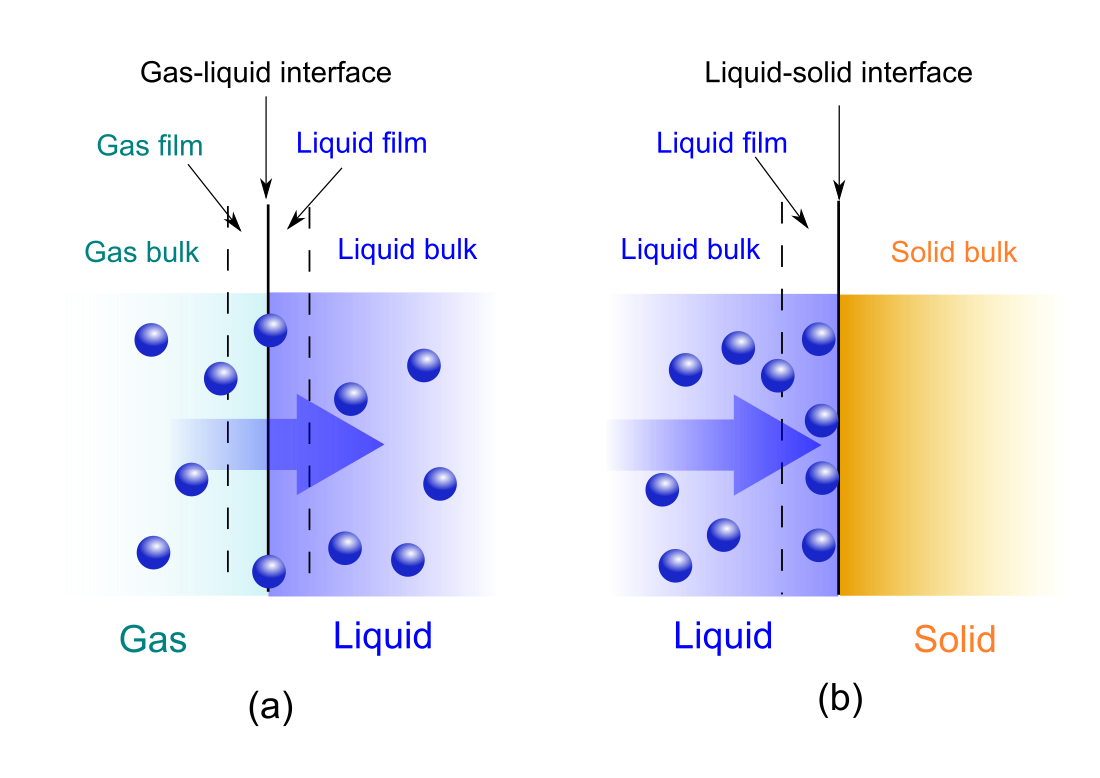
The adsorption and absorption rate of a diluted solute in gas or liquid solution to a surface or interface can be calculated using Fick's laws of diffusion.
See also
References
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.