Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
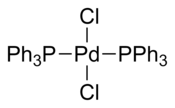
Bis(triphenylphosphine)palladium chloride is a coordination compound of palladium containing two triphenylphosphine and two chloride ligands. It is a yellow solid that is soluble in some organic solvents. It is used for palladium-catalyzed coupling reactions, e.g. the Sonogashira–Hagihara reaction. The complex is square planar. Many analogous complexes are known with different phosphine ligands.
 | |
 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.034.299 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| PdCl2(PPh3)2 | |
| Molar mass | 701.90 g·mol−1 |
| Appearance | yellow powder |
| Melting point | 260 °C (decomposed around 300 °C) |
| insoluble | |
| Solubility | soluble in chloroform, hexane, toluene, benzene, acetone[1] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 181.7 °C |
| Related compounds | |
Related compounds |
Bis(triphenylphosphine)platinum chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
This compound may be prepared by treating palladium(II) chloride with triphenylphosphine:[2][3]
Upon reduction with hydrazine in the presence of excess triphenylphosphine, the complex is a precursor to tetrakis(triphenylphosphine)palladium, Pd(PPh3)4:[4]
Several crystal structures containing PdCl2(PPh3)2 have been reported. In all of the structures, PdCl2(PPh3)2 adopts a square planar coordination geometry and the trans isomeric form.[5][6][7][8]
The complex is used as a pre-catalyst for a variety of coupling reactions.[9]

The Suzuki reaction was once limited by high levels of catalyst and the limited availability of boronic acids. Replacements for halides were also found, increasing the number of coupling partners for the halide or pseudohalide as well. Using bis(triphenylphosphine)palladium chloride as the catalyst, triflates and boronic acids have been coupled on an 80 kilogram scale in good yield.[10] The same catalyst is effective for the Sonogashira coupling.[11]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.