Loading AI tools
Pair of stereoisomers From Wikipedia, the free encyclopedia
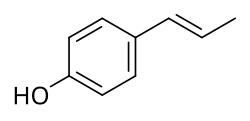
Anol, also known as p-hydroxypropenylbenzene,[1] is a simple phenol that was derived via demethylation from anethole, an estrogenic constituent of anise and fennel, by Sir Charles Dodds in 1937.[2][3] It was reported to possess extremely potent estrogenic activity on par with that of steroidal estrogens like estrone, with a dose of 1 μg inducing estrus in rats.[2] However, subsequent studies with different preparations of anol failed to confirm these findings, and it was found that dimerization of anol into dianol and hexestrol can rapidly occur and that the latter impurity was responsible for the highly potent estrogenic effects.[4] [2][3][5][6] Dodds later synthesized the structurally related and extremely potent estrogen diethylstilbestrol in 1938.[2][5]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C9H10O |
| Molar mass | 134.178 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.