Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
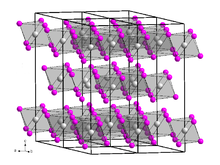
Vanadium(III) bromide, also known as vanadium tribromide, describes the inorganic compounds with the formula VBr3 and its hydrates. The anhydrous material is a green-black solid. In terms of its structure, the compound is polymeric with octahedral vanadium(III) surrounded by six bromide ligands.
 | |
| Names | |
|---|---|
| IUPAC name
Vanadium(III) bromide | |
| Other names
Vanadium tribromide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.382 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| VBr3 | |
| Molar mass | 290.654 g/mol |
| Appearance | Green-black solid |
| Density | 4 g/cm3 |
| soluble | |
| Solubility | soluble in THF (forms adduct) |
| +2890.0·10−6 cm3/mol | |
| Structure | |
| octahedral | |
| Related compounds | |
Other anions |
Vanadium(III) chloride |
Other cations |
Titanium(III) bromide Molybdenum(III) bromide |
Related compounds |
Vanadium(II) bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
VBr3 has been prepared by treatment of vanadium tetrachloride with hydrogen bromide:
The reaction proceeds via the unstable vanadium(IV) bromide (VBr4), which releases Br2 near room temperature.[1]
It is also possible to prepare vanadium(III) bromide by reacting bromine with vanadium or ferrovanadine:[2]
Vanadium(III) bromide is present in the form of black, leafy, very hygroscopic crystals with a sometimes greenish sheen. It is soluble in water with green color. Its crystal structure is isotypic to that of vanadium(III) chloride with space group R3c (space group no. 167), a = 6.400 Å, c = 18.53 Å. When heated to temperatures of around 500 °C, a violet gas phase is formed, from which, under suitable conditions, red vanadium(IV) bromide can be separated by rapid cooling, which decomposes at −23 °C.[2]
Like vanadium(III) chloride, vanadium(III) bromide forms red-brown soluble complexes with dimethoxyethane and THF, such as mer-VBr3(THF)3.[3]
Aqueous solutions prepared from VBr3 contain the cation trans-[VBr2(H2O)4]+. Evaporation of these solutions give the salt trans-[VBr2(H2O)4]Br.(H2O)2.[4]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.