Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
Vanadium(II) fluoride is a fluoride of vanadium, with the chemical formula of VF2. It forms blue crystals.
 | |
| Names | |
|---|---|
| Other names
Vanadium(II) fluoride | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| F2V | |
| Molar mass | 88.9383 g·mol−1 |
| Appearance | blue crystals |
| soluble in water, forms [V(H2O)6]2+[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Vanadium(II) fluoride can be produced by the reduction of vanadium trifluoride by hydrogen in a hydrogen fluoride atmosphere at 1150 °C:[3]
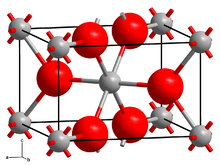
Vanadium(II) fluoride crystallizes in the tetragonal crystal system with space group P42/mnm (No. 136). Its lattice constants are a = 480.4 pm and c = 323.7 pm.[4]
Vanadium(II) fluoride is a strong reducing agent that can reduce nitrogen to hydrazine in the presence of magnesium hydroxide.[2]
It dissolves in water to form [V(H2O)6]2+ ions.[2]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.