User:Isabel.guillen.5/sandbox
Chemical compound / From Wikipedia, the free encyclopedia
Quercetin is the most common flavonoid in nature. It is found as its glycosylated forms such as quercitrin (3-rhamnosylquercetin) or rutoside (3-rhamnosy-glucosyl quercetin),[2] specifically as a flavonol, which is a polyphenolic compound synthesized in plant cells.[3] It exhibits antioxidant properties by reducing oxidative stress in cells via two distinct pathways: (1) radical scavenging activities and binding of transition metal ions and (2) competitively inhibiting xanthine oxidase (XOD).[4] Quercetin is familiarly used in traditional medicine to prevent or treat diseases such as cancer, cardiovascular and nervous diseases, obesity, and chronic inflammation. Quercetin is naturally found in fruits and vegetables, especially onions, citrus and apples. Other sources include dark berries, grapes and olive oil. Green tea and red wine because of their high percentage of antioxidants and flavonoids have been identified as having prominent amounts of quercetin. [5]
 | This is the user sandbox of Isabel.guillen.5. A user sandbox is a subpage of the user's user page. It serves as a testing spot and page development space for the user and is not an encyclopedia article. Create or edit your own sandbox here. Other sandboxes: Main sandbox | Template sandbox Finished writing a draft article? Are you ready to request review of it by an experienced editor for possible inclusion in Wikipedia? Submit your draft for review! |
 | |
| Names | |
|---|---|
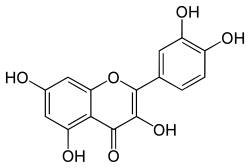
| IUPAC name
2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chromen-4-one | |
| Other names
Sophoretin Meletin Quercetine Xanthaurine Quercetol Quercitin Quertine Flavin meletin | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL |
|
| ChemSpider |
|
| DrugBank |
|
| KEGG |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C15H10O7 | |
| Molar mass | 302.236 g/mol |
| Appearance | yellow crystalline powder[1] |
| Density | 1.799 g/cm3 |
| Melting point | 316 °C |
| Practically insoluble in water; soluble in aqueous alkaline solutions[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
