
tert-Butyllithium
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tert-Butyllithium?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
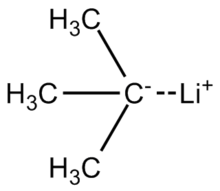
tert-Butyllithium is a chemical compound with the formula (CH3)3CLi. As an organolithium compound, it has applications in organic synthesis since it is a strong base, capable of deprotonating many carbon molecules, including benzene. tert-Butyllithium is available commercially as solutions in hydrocarbons (such as pentane); it is not usually prepared in the laboratory.
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
| |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3587204 | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.939 |
| EC Number |
|
PubChem CID |
|
| UN number | 3394 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LiC 4H 9 | |
| Molar mass | 64.055 g mol−1 |
| Appearance | Colorless solid |
| Density | 660 mg cm−3 |
| Boiling point | 36 to 40 °C (97 to 104 °F; 309 to 313 K) |
| Reacts | |
| Acidity (pKa) | 45–53 |
| Hazards | |
| GHS labelling: | |
     | |
| Danger | |
| H225, H250, H260, H300, H304, H310, H314, H330, H336, H411 | |
| P210, P222, P223, P231+P232, P370+P378, P422 | |
| NFPA 704 (fire diamond) | |
| Flash point | −6.6 °C (20.1 °F; 266.5 K) |
| Related compounds | |
Related compounds |
n-Butyllithium |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
