
Creatinine
Breakdown product of creatine phosphate / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Serum creatinine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with creatine or Creatine kinase.
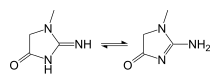
Creatinine (/kriˈætɪnɪn, -niːn/; from Ancient Greek: κρέας (kréas) 'flesh') is a breakdown product of creatine phosphate from muscle and protein metabolism. It is released at a constant rate by the body (depending on muscle mass).[3][4]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
| |
| Other names | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 3DMet | |
| 112061 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank | |
| ECHA InfoCard | 100.000.424 |
| EC Number |
|
| KEGG |
|
| MeSH | Creatinine |
PubChem CID |
|
| UNII | |
| UN number | 1789 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H7N3O | |
| Molar mass | 113.120 g·mol−1 |
| Appearance | White crystals |
| Density | 1.09 g cm−3 |
| Melting point | 300 °C (572 °F; 573 K)[1] (decomposes) |
| 1 part per 12[1]
90 mg/mL at 20°C[2] | |
| log P | -1.76 |
| Acidity (pKa) | 12.309 |
| Basicity (pKb) | 1.688 |
| Isoelectric point | 11.19 |
| Thermochemistry | |
Heat capacity (C) |
138.1 J K−1 mol−1 (at 23.4 °C) |
Std molar entropy (S⦵298) |
167.4 J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) |
−240.81–239.05 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−2.33539–2.33367 MJ mol−1 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 290 °C (554 °F; 563 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
