Loading AI tools
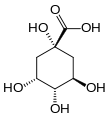
Quinic acid is a cyclitol, a cyclic polyol, and a cyclohexanecarboxylic acid. It is a colorless solid that can be extracted from plant sources. Quinic acid is implicated in the perceived acidity of coffee.
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
(1S,3R,4S,5R)-1,3,4,5-Tetrahydroxycyclohexane-1-carboxylic acid | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.976 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C7H12O6 | |||
| Molar mass | 192.17 g/mol | ||
| Density | 1.35 g/cm3 | ||
| Melting point | 168 °C (334 °F; 441 K) | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
The compound is obtained from cinchona bark, coffee beans, and the bark of Eucalyptus globulus.[1] It is a constituent of the tara tannins.
Urtica dioica, the European stinging nettle, is another common source.[citation needed]
It is made synthetically by hydrolysis of chlorogenic acid. Quinic acid is also implicated in the perceived acidity of coffee.

This substance was isolated for the first time in 1790 by German pharmacist Friedrich Christian Hofmann in Leer from Cinchona.[2] Its transformation into hippuric acid by animal metabolism was studied by German chemist Eduard Lautemann in 1863.[3]
Its biosynthesis begins with the transformation of glucose into erythrose 4-phosphate. This four-carbon substrate is condensed with phosphoenol pyruvate to give the seven-carbon 3-deoxy-D-arabinoheptulosonate 7-phosphate (DAHP) by the action of a synthase. Two subsequent steps involving dehydroquinic acid synthase and a dehydrogenase afford the compound.[4]
Derived bicyclic lactones are called quinides. One example is 4-caffeoyl-1,5-quinide.
Dehydrogenation and oxidation of quinic acid affords gallic acid.[4]
Quinic acid is used as an astringent.
This acid is a versatile chiral starting material for the synthesis of pharmaceuticals.[4] It is a building block in the synthesis of Oseltamivir, which is used to treat influenza A and B.
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.