
Lithium cobalt oxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lithium Cobalt Oxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
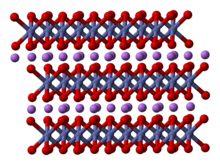
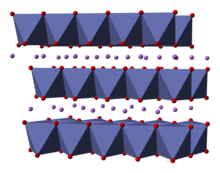
Lithium cobalt oxide, sometimes called lithium cobaltate[2] or lithium cobaltite,[3] is a chemical compound with formula LiCoO
2. The cobalt atoms are formally in the +3 oxidation state, hence the IUPAC name lithium cobalt(III) oxide.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
lithium cobalt(III) oxide | |
| Other names
lithium cobaltite | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.032.135 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LiCoO 2 | |
| Molar mass | 97.87 g mol−1 |
| Appearance | dark blue or bluish-gray crystalline solid |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
harmful |
| GHS labelling: | |
  | |
| Danger | |
| H317, H350, H360 | |
| P201, P202, P261, P272, P280, P281, P302+P352, P308+P313, P321, P333+P313, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Lithium cobalt oxide is a dark blue or bluish-gray crystalline solid,[4] and is commonly used in the positive electrodes of lithium-ion batteries.