
Lithium bis(trimethylsilyl)amide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about LiHMDS?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
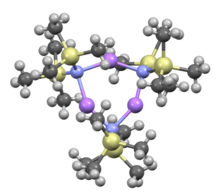
Lithium bis(trimethylsilyl)amide is a lithiated organosilicon compound with the formula LiN(Si(CH3)3)2. It is commonly abbreviated as LiHMDS or Li(HMDS) (lithium hexamethyldisilazide - a reference to its conjugate acid HMDS) and is primarily used as a strong non-nucleophilic base and as a ligand. Like many lithium reagents, it has a tendency to aggregate and will form a cyclic trimer in the absence of coordinating species.
Quick Facts Names, Identifiers ...
 Monomer (does not exist) | |
 Cyclic trimer | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Lithium 1,1,1-trimethyl-N-(trimethylsilyl)silanaminide | |
| Other names
Lithium hexamethyldisilazide Hexamethyldisilazane lithium salt | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.021.569 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LiN(Si(CH3)3)2 | |
| Molar mass | 167.33 g·mol−1 |
| Appearance | White solid |
| Density | 0.86 g/cm3 at 25 °C |
| Melting point | 71 to 72 °C (160 to 162 °F; 344 to 345 K) |
| Boiling point | 80 to 84 °C (176 to 183 °F; 353 to 357 K) (0.001 mm Hg) |
| decomposes | |
| Solubility | Most aprotic solvents THF, hexane, toluene |
| Acidity (pKa) | 26 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
flammable, corrosive |
| Related compounds | |
Related compounds |
Sodium bis(trimethylsilyl)amide Potassium bis(trimethylsilyl)amide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close