Lamotrigine
Medication used for bipolar disorder, epilepsy, & many seizure disorders / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lamictal?
Summarize this article for a 10 year old
Lamotrigine, sold under the brand name Lamictal among others, is a medication used to treat epilepsy and stabilize mood in bipolar disorder.[5][8] For epilepsy, this includes focal seizures, tonic-clonic seizures, and seizures in Lennox-Gastaut syndrome.[8] In bipolar disorder, lamotrigine has not been shown to reliably treat acute depression in any groups except for the severely depressed; but for patients with bipolar disorder who are not currently symptomatic, it appears to reduce the risk of future episodes of depression.[9]
| |||
| Clinical data | |||
|---|---|---|---|
| Pronunciation | /ləˈmoʊtrɪˌdʒiːn/ | ||
| Trade names | Lamictal, others[1] | ||
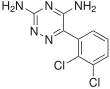
| Other names | BW-430C; BW430C; 3,5-Diamino-6-(2,3-dichlorophenyl)-1,2,4-triazine | ||
| AHFS/Drugs.com | Monograph | ||
| MedlinePlus | a695007 | ||
| License data |
| ||
| Pregnancy category |
| ||
| Routes of administration | Oral (by mouth) | ||
| Drug class | Phenyltriazine | ||
| ATC code | |||
| Legal status | |||
| Legal status | |||
| Pharmacokinetic data | |||
| Bioavailability | 98% | ||
| Protein binding | 55% | ||
| Metabolism | Liver (mostly UGT1A4-mediated) | ||
| Elimination half-life | 29 hours | ||
| Excretion | Urine (65%), feces (2%) | ||
| Identifiers | |||
| |||
| CAS Number | |||
| PubChem CID | |||
| IUPHAR/BPS | |||
| DrugBank |
| ||
| ChemSpider |
| ||
| UNII | |||
| KEGG |
| ||
| ChEBI | |||
| ChEMBL | |||
| CompTox Dashboard (EPA) | |||
| ECHA InfoCard | 100.074.432 | ||
| Chemical and physical data | |||
| Formula | C9H7Cl2N5 | ||
| Molar mass | 256.09 g·mol−1 | ||
| 3D model (JSmol) | |||
| |||
| |||
| | |||
Common side effects include nausea, sleepiness, headache, vomiting, trouble with coordination, and rash.[8] Serious side effects include excessive breakdown of red blood cells, increased risk of suicide, severe skin reaction (Stevens–Johnson syndrome), and allergic reactions, which can be fatal.[8] Lamotrigine is a phenyltriazine,[5] making it chemically different from other anticonvulsants.[8] Its mechanism of action is not clear, but it appears to inhibit release of excitatory neurotransmitters via voltage-sensitive sodium channels and voltage-gated calcium channels in neurons.[8][10][11]
Lamotrigine was first marketed in Ireland in 1991,[12] and approved for use in the United States in 1994.[8][13] It is on the World Health Organization's List of Essential Medicines.[14] In 2021, it was the 50th most commonly prescribed medication in the United States, with more than 13 million prescriptions.[15][16]