File:Bupropion_bioequivalency_comparison.svg
From Wikipedia, the free encyclopedia
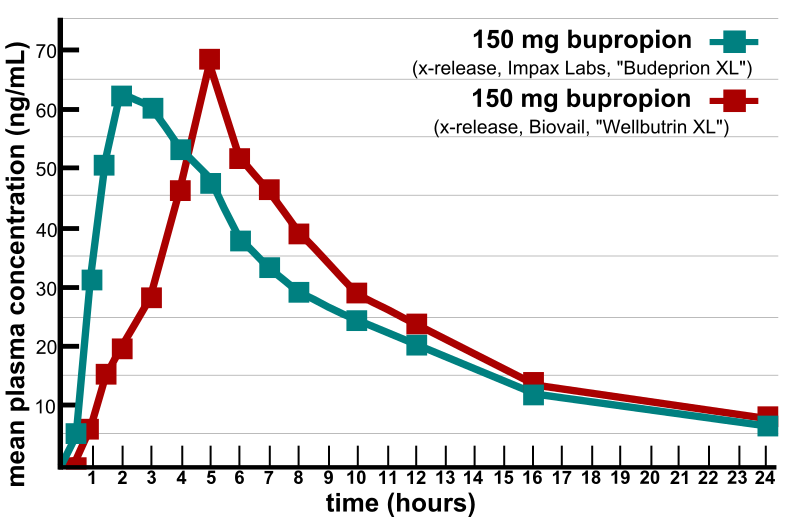
Size of this PNG preview of this SVG file: 800 × 520 pixels. Other resolutions: 320 × 208 pixels | 640 × 416 pixels | 1,024 × 666 pixels | 1,280 × 832 pixels | 2,560 × 1,664 pixels | 1,000 × 650 pixels.
Original file (SVG file, nominally 1,000 × 650 pixels, file size: 260 KB)
| This is a file from the Wikimedia Commons. Information from its description page there is shown below. Commons is a freely licensed media file repository. You can help. |
Contents
Summary
| DescriptionBupropion bioequivalency comparison.svg |
A bioequivalency profile comparison of extended-release bupropion as produced by Impax Laboratories for Teva and Biovail for GlaxoSmithKline. Note: The generic drug illustrated in this graphic was not affected by the FDA's 300 mg recall and is still available in the United States as of April 2013.[1][2] |
| Date | |
| Source | Own work based on FDA data [3] |
| Author | CMBJ |
Licensing
I, the copyright holder of this work, hereby publish it under the following license:
This file is licensed under the Creative Commons Attribution 3.0 Unported license.
- You are free:
- to share – to copy, distribute and transmit the work
- to remix – to adapt the work
- Under the following conditions:
- attribution – You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
See also
Notes
- ↑ (2012-10-03). "FDA Update: Budeprion XL 300 mg Not Therapeutically Equivalent to Wellbutrin XL 300 mg". Food and Drug Administration. Retrieved on 2013-04-29.
- ↑ Moeller, Karen E.. "Wellbutrin/Budeprion XL 300 mg". NAMI. Retrieved on 2013-04-28.
- ↑
Time
(hours)Mean plasma concentration, Impax
(ng/mL)Mean plasma concentration, Biovail
(ng/mL)0 0 0 0.5 5 1 1 31.5 6.5 1.5 51 15 2 62 19.5 3 60 28 4 53 46 5 48 67 6 38 51.5 7 33 46 8 29 39 10 24 28.5 12 20 23.5 16 12 13.5 24 6.5 8 Data derived from FDA graphic chart.
Captions
Add a one-line explanation of what this file represents
Items portrayed in this file
depicts
28 April 2013
image/svg+xml
ec11e6dd6b0e5ca8e49e8fd1d1d4bdeaf0da0712
266,229 byte
650 pixel
1,000 pixel
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 08:59, 29 April 2013 |  | 1,000 × 650 (260 KB) | CMBJ | |
| 02:18, 29 April 2013 |  | 1,000 × 650 (180 KB) | CMBJ | ||
| 08:33, 28 April 2013 |  | 700 × 500 (32 KB) | CMBJ | ||
| 05:58, 28 April 2013 |  | 1,000 × 500 (41 KB) | CMBJ | ||
| 04:35, 28 April 2013 |  | 2,100 × 1,000 (39 KB) | CMBJ | {{Information |Description=Note: The generic drug illustrated in this graphic was not affected by the FDA's recall and is still available in the United States as of April 2013.<ref name='Moeller'> {{cite news | first = Karen E. | last = Moeller | title... |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on ar.wikipedia.org
- Usage on es.wikipedia.org
- Usage on fi.wikipedia.org
- Usage on fr.wikipedia.org
- Usage on id.wikipedia.org
- Usage on ja.wikipedia.org
- Usage on lv.wikipedia.org
- Usage on pl.wikipedia.org
- Usage on sh.wikipedia.org
- Usage on sr.wikipedia.org
- Usage on th.wikipedia.org
- Usage on zh.wikipedia.org
Metadata
This file contains additional information, probably added from the digital camera or scanner used to create or digitize it.
If the file has been modified from its original state, some details may not fully reflect the modified file.
| Width | 1000 |
|---|---|
| Height | 650 |