
Charles's law
Relationship between volume and temperature of a gas at constant pressure / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Charles's law?
Summarize this article for a 10 year old
Charles' law (also known as the law of volumes) is an experimental gas law that describes how gases tend to expand when heated. A modern statement of Charles' law is:
When the pressure on a sample of a dry gas is held constant, the Kelvin temperature and the volume will be in direct proportion.[1]

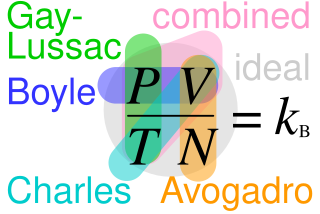
This relationship of direct proportion can be written as:
So this means:
where:
- V is the volume of the gas,
- T is the temperature of the gas (measured in kelvins), and
- k is a non-zero constant.
This law describes how a gas expands as the temperature increases; conversely, a decrease in temperature will lead to a decrease in volume. For comparing the same substance under two different sets of conditions, the law can be written as:
The equation shows that, as absolute temperature increases, the volume of the gas also increases in proportion.


