Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
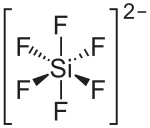
Barium hexafluorosilicate is an inorganic chemical compound with the chemical formula BaSiF6.[3][4][5]
 | |
| Names | |
|---|---|
| IUPAC name
barium(2+);hexafluorosilicon(2-) | |
| Other names
Barium silicofluoride, bariumsilicofluorid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.037.430 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| BaF6Si | |
| Molar mass | 279.402 g·mol−1 |
| Appearance | White crystalline powder |
| Density | 4.279 g/cm3[1] |
| Melting point | 1580 |
| poorly soluble | |
| Hazards | |
| GHS labelling:[2] | |
 | |
| Warning | |
| H302, H332 | |
| P261, P264, P270, P271, P301+P317, P304+P340, P317, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
As a salt that is poorly soluble in water, barium hexafluorosilicate precipitates from solutions that contain barium ions (e.g. barium chloride as well as hexafluorosilicate ions (e.g. hexafluorosilicic acid).[6]
The compound is used as a chemical reagent in experimental applications. In various chemical reactions and processes, the compound acts as a source of barium and hexafluorosilicate ions.[7]
It was also used as an insecticide.[8]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.