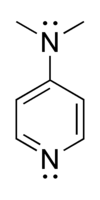
4-Dimethylaminopyridine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about 4-dimethylaminopyridine?
Summarize this article for a 10 year old
4-Dimethylaminopyridine (DMAP) is a derivative of pyridine with the chemical formula (CH3)2NC5H4N. This white solid is of interest because it is more basic than pyridine, owing to the resonance stabilisation from the NMe2 substituent.
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
N,N-Dimethylpyridin-4-amine Dimethyl(pyridin-4-yl)azane Dimethyl(pyridin-4-yl)amine | |||
| Preferred IUPAC name
N,N-Dimethylpyridin-4-amine | |||
| Other names
4-(Dimethylamino)pyridine N,N-Dimethyl-4-aminopyridine DMAP 4-Dimethylaminopyridine 4-(Dimethylamino)azine N,N-dimethyl-4-aminoazine 4-(Dimethylamino)azabenzene N,N-Dimethyl-4-aminoazabenzene | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider |
| ||
| ECHA InfoCard | 100.013.049 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C7H10N2 | |||
| Molar mass | 122.17 g/mol | ||
| Appearance | white solid | ||
| Melting point | 110 to 113 °C (230 to 235 °F; 383 to 386 K) | ||
| Boiling point | 162 °C (324 °F; 435 K) at 50 mmHg | ||
| Acidity (pKa) | 9.6 in water, 17.95 (pKa of conjugate acid in acetonitrile)[1] | ||
| Hazards | |||
| GHS labelling: | |||
 | |||
| Danger | |||
| H301, H310, H315, H319, H335[2] | |||
| P280, P305+P351+P338, P337+P313[2] | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
deer mice: oral, 450 mg/kg[3] mice: oral, 350 mg/kg/day[3] | ||
| Safety data sheet (SDS) | [2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Because of its basicity, DMAP is a useful nucleophilic catalyst for a variety of reactions such as esterifications with anhydrides, the Baylis-Hillman reaction, hydrosilylations, tritylation, the Steglich rearrangement, Staudinger synthesis of β-lactams and many more. Chiral DMAP analogues are used in kinetic resolution experiments of mainly secondary alcohols and Evans auxiliary type amides.[4][5][6]