Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
1,1'-Azobis-1,2,3-triazole is a moderately explosive but comparatively stable chemical compound[citation needed] which contains a long continuous chain of nitrogen atoms, with an unbroken chain of eight nitrogen atoms cyclised into two 1,2,3-triazole rings. It is stable up to 194 °C. The compound exhibits cis–trans isomerism at the central azo group: the trans isomer is more stable and is yellow, while the cis isomer is less stable and is blue. The two rings are aromatic and form a conjugated system with the azo linkage. This chromophore allows the trans compound to be isomerised to the cis when treated with an appropriate wavelength of ultraviolet light.[1]

 | |
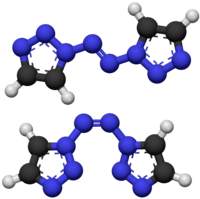 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′-Diazenediyldi(1H-1,2,3-triazole) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C4H4N8 | |
| Molar mass | 164.132 g·mol−1 |
| Appearance | Blue (cis isomer) Yellow (trans isomer) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Explosive |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In 2011, 1,1'-azobis(1,2,3,4-tetrazole) with formula C2H2N10 was prepared by Klapötke and Piercey which has a 10-nitrogen chain.[2] The record was later taken by a 11-nitrogen chain compound synthesized by a group of Chinese researchers.[3][which?] A branched chain of 11-nitrogen system has also been reported as part of an unstable but highly nitrogen rich azidotetrazole derivative with formula C2N14.[4]

Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.