
Trigonal bipyramidal molecular geometry
Molecular structure with atoms at the center and vertices of a triangular bipyramid / From Wikipedia, the free encyclopedia
In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid.[1] This is one geometry for which the bond angles surrounding the central atom are not identical (see also pentagonal bipyramid), because there is no geometrical arrangement with five terminal atoms in equivalent positions. Examples of this molecular geometry are phosphorus pentafluoride (PF5), and phosphorus pentachloride (PCl5) in the gas phase.[2]
Quick Facts Examples, Point group ...
| Trigonal bipyramidal molecular geometry | |
|---|---|
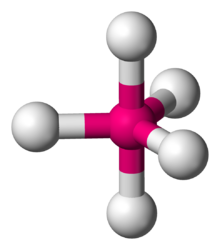 | |
| Examples | PF5, Fe(CO)5 |
| Point group | D3h |
| Coordination number | 5 |
| Bond angle(s) | 90°, 120° |
| μ (Polarity) | 0 |
Close