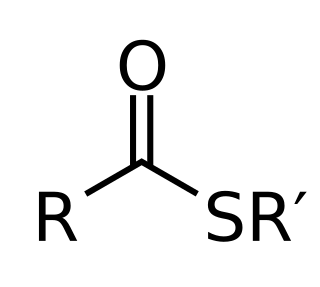
Thioester
Organosulfur compounds of the form R–SC(=O)–R’ / From Wikipedia, the free encyclopedia
In organic chemistry, thioesters are organosulfur compounds with the molecular structure R−C(=O)−S−R’. They are analogous to carboxylate esters (R−C(=O)−O−R’) with the sulfur in the thioester replacing oxygen in the carboxylate ester, as implied by the thio- prefix. They are the product of esterification of a carboxylic acid (R−C(=O)−O−H) with a thiol (R'−S−H). In biochemistry, the best-known thioesters are derivatives of coenzyme A, e.g., acetyl-CoA.[1] The R and R' represent organyl groups, or H in the case of R.