
Enantiomer
Stereoisomers that are nonsuperposable mirror images of each other / From Wikipedia, the free encyclopedia
In chemistry, an enantiomer (/ɪˈnænti.əmər, ɛ-, -oʊ-/[1] ih-NAN-tee-ə-mər; from Ancient Greek ἐναντίος (enantíos) 'opposite', and μέρος (méros) 'part') – also called optical isomer,[2] antipode,[3] or optical antipode[4] – is one of two stereoisomers that are nonsuperposable onto their own mirror image. Enantiomers of each other are much like one's right and left hands; without mirroring one of them, hands cannot be superposed onto each other.[5] It is solely a relationship of chirality and the permanent three-dimensional relationships among molecules or other chemical structures: no amount of re-orientiation of a molecule as a whole or conformational change converts one chemical into its enantiomer. Chemical structures with chirality rotate plane-polarized light.[6] A mixture of equal amounts of each enantiomer, a racemic mixture or a racemate, does not rotate light.[7][8][9]
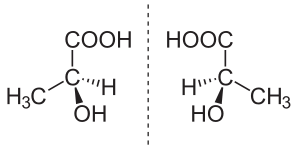
Stereoisomers include both enantiomers and diastereomers. Diastereomers, like enantiomers, share the same molecular formula and are also nonsuperposable onto each other; however, they are not mirror images of each other.[10]