
Carboxylate
Chemical group (RCOO); conjugate base of a carboxylic acid / From Wikipedia, the free encyclopedia
In organic chemistry, a carboxylate is the conjugate base of a carboxylic acid, RCOO− (or RCO−2). It is an ion with negative charge.

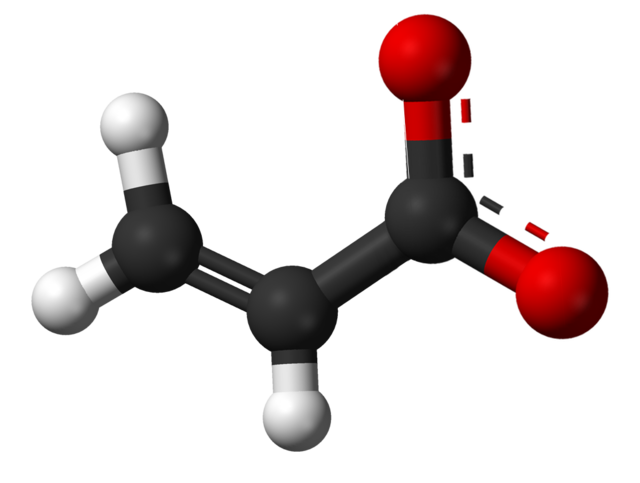
Carboxylate salts are salts that have the general formula M(RCOO)n, where M is a metal and n is 1, 2,.... Carboxylate esters have the general formula RCOOR′ (also written as RCO2R′), where R and R′ are organic groups.