
Thiazide
Class of chemical compounds / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about thiazide?
Summarize this article for a 10 year old
Thiazide (/ˈθaɪəzaɪd/) refers to both a class of sulfur-containing organic molecules[1] and a class of diuretics based on the chemical structure of benzothiadiazine.[2] The thiazide drug class was discovered and developed at Merck and Co. in the 1950s.[3] The first approved drug of this class, chlorothiazide, was marketed under the trade name Diuril beginning in 1958.[3] In most countries, thiazides are the least expensive antihypertensive drugs available.[4]
| Thiazide | |
|---|---|
| Drug class | |
 Chlorothiazide, the first thiazide drug | |
| Class identifiers | |
| Use | hypertension, edema |
| ATC code | C03A |
| Biological target | sodium-chloride symporter |
| External links | |
| MeSH | D049971 |
| Legal status | |
| In Wikidata | |
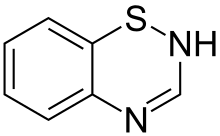
Thiazide organic molecules are bi-cyclic structures that contain adjacent sulfur and nitrogen atoms on one ring.[5] Confusion sometimes occurs because thiazide-like diuretics such as indapamide are referred to as thiazides despite not having the thiazide chemical structure.[6] When used this way, "thiazide" refers to a drug which acts at the thiazide receptor.[7] The thiazide receptor is a sodium-chloride transporter that pulls NaCl from the lumen in the distal convoluted tubule. Thiazide diuretics inhibit this receptor, causing the body to release NaCl and water into the lumen, thereby increasing the amount of urine produced each day.[6] An example of a molecule that is chemically a thiazide but not used as a diuretic is methylchloroisothiazolinone, often found as an antimicrobial in cosmetics.[8]