Dissociation (chemistry)
Separation of molecules or ionic compounds into smaller constituent entities / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dissociate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Dissociate" and "Dissociating" redirect here. For other uses, see Dissociation.
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salts, or complexes) separate or split into other things such as atoms, ions, or radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination.
This article needs additional citations for verification. (June 2014) |
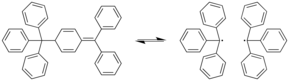
Dissociation of Gomberg's dimer
Dissociation diagram of phosphoric acid