
Neoprene
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Chloroprene rubber?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Neoprene (also polychloroprene) is a family of synthetic rubbers that are produced by polymerization of chloroprene.[1] Neoprene exhibits good chemical stability and maintains flexibility over a wide temperature range. Neoprene is sold either as solid rubber or in latex form and is used in a wide variety of commercial applications, such as laptop sleeves, orthopaedic braces (wrist, knee, etc.), electrical insulation, medical gloves, liquid and sheet-applied elastomeric membranes or flashings, and automotive fan belts.[2]
Quick Facts Identifiers, Properties ...
 A neck seal, wrist seal, manual vent, inflator, zip and fabric of a neoprene dry suit. The soft seal material at the neck and wrists is made from single backed closed-cell foam neoprene for elasticity. The slick unbacked side seals against the skin. The blue area is double-backed with knit nylon fabric laminated onto closed cell foamed neoprene for toughness. Some insulation is provided by the suit, and the rest by garments worn underneath. | |
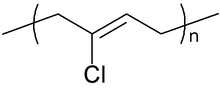 Chemical structure of the repeating unit of polychloroprene | |
| Identifiers | |
|---|---|
| ECHA InfoCard | 100.127.980 |
| EC Number |
|
CompTox Dashboard (EPA) |
|
| Properties | |
| Density | 1.23 g/cm3 (solid) 0.1-0.3 g/cm3 (foam) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close